Necrotizing enterocolitis is the most common and most deadly gastrointestinal emergency in preterm infants. It destroys intestinal tissue, causes sepsis, and kills between 20 and 30 percent of the babies who develop it. For the survivors, the consequences often include short bowel syndrome, lifelong nutritional challenges, and neurodevelopmental impairment.
The standard explanation focuses on bacterial colonization in an immature gut. The standard interventions focus on antibiotics, surgical resection, and feeding protocols. These save some lives. They do not address why certain bacteria become pathogenic in the preterm gut in the first place.
I believe the answer involves nickel. Specifically, dietary nickel in infant formula activating nickel-dependent virulence pathways in bacteria that are already present in the preterm gut.
The mechanism
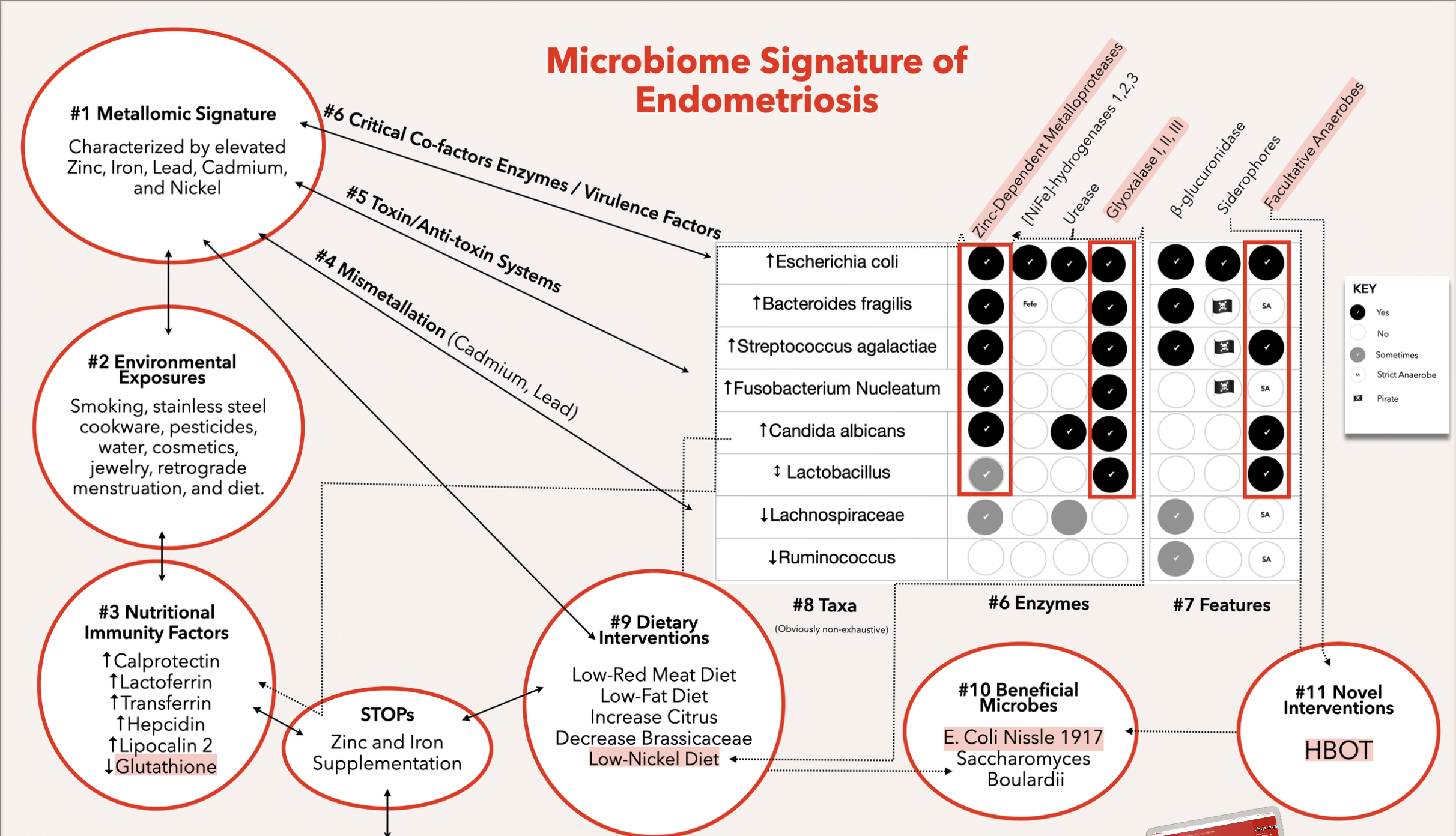
The bacteria most commonly implicated in NEC, particularly pathogenic strains of Escherichia coli, carry virulence factors that depend on nickel as an essential cofactor. These include urease, which allows bacteria to survive acid stress and colonize hostile environments. [NiFe]-hydrogenase, which provides metabolic energy under anaerobic conditions. And Ni-glyoxalase I, which detoxifies methylglyoxal produced during inflammation, enabling bacteria to survive the host's immune response.
None of these enzymes require nickel from the host's perspective. The human body does not use nickel-dependent urease, [NiFe]-hydrogenase, or Ni-glyoxalase. This creates an asymmetry: nickel in the diet benefits pathogenic bacteria while providing nothing to the infant.
The preterm gut is uniquely vulnerable to this asymmetry. The mucosal barrier is immature. The immune system is underdeveloped. The microbiome is sparse and unstable, easily shifted by environmental inputs. And the primary environmental input is formula.
Infant formula contains nickel. The amount varies by product, by manufacturing process, by water source, and by packaging material. There are no regulatory limits specific to nickel in infant formula. There are no testing requirements. There is no certification standard.
When nickel enters the preterm gut, it becomes bioavailable to bacteria carrying nickel-dependent virulence genes. These bacteria now have the cofactor they need to activate urease (colonization advantage), [NiFe]-hydrogenase (metabolic advantage), and Ni-glyoxalase (immune evasion advantage). The result is a pathobiont with a full virulence toolkit, operating in a gut that lacks the barriers and immune capacity to contain it.
The host's primary defense against this, nutritional immunity, is the strategy of sequestering metals away from pathogens. Neutrophils release calprotectin, a protein that binds zinc and nickel with high affinity, starving bacteria of these cofactors. But preterm infants have limited neutrophil function and reduced calprotectin production. The nutritional immunity system that would normally constrain nickel-dependent virulence is not yet operational.
The mechanism, from start to finish: dietary nickel enters the preterm gut through formula, activates nickel-dependent virulence pathways in colonizing pathobionts, overwhelms an immature nutritional immunity system, and triggers the inflammatory cascade that becomes necrotizing enterocolitis.
I published the full framework in a paper on Zenodo earlier this year.
Why this matters beyond the NICU
The NEC/nickel hypothesis is a specific instance of a broader pattern that I've been researching across multiple conditions through the field of microbial metallomics. Heavy metals don't just poison host cells. They reshape the microbial ecosystem by providing virulence-enabling cofactors to pathogenic bacteria while the host derives no benefit from their presence.
This pattern appears in Parkinson's disease, where iron dyshomeostasis selects for metal-resistant gut pathobionts. It appears in endometriosis, where nickel-dependent virulence factors enable pathobiont persistence in inflamed pelvic tissue. And it appears in the preterm gut, where nickel in formula may be the catalytic input that converts a colonizing bacterium into a pathogen.
The World Health Organization's top priority pathogens, the bacteria they are most concerned about in terms of antibiotic resistance, are all nickel-dependent for their virulence factors. This is not a coincidence. It is a signal that nickel contamination in the food supply has consequences far beyond what current toxicology models account for.
What can be done
Three things.
First, test infant formula for nickel. Not just lead, arsenic, cadmium, and mercury (which are increasingly tested under regulatory pressure), but nickel specifically. The analytical methods exist. The labs exist. What doesn't exist is the requirement, the standard, or the consumer awareness that would drive testing.
This is one of the reasons I built HMTc (Heavy Metal Tested and Certified). The HMTc standards cover eight metals including nickel, with category-specific contaminant limits, ALARA-based principles, and lot testing schedules. Infant food is one of the priority categories. The infrastructure for testing and certifying infant formula for nickel content exists today through HMTc. What is needed is adoption.
Second, fund research on nickel speciation in infant formula and its bioavailability to gut bacteria. The published literature on nickel in infant formula is thin. We know nickel is present. We do not know in what chemical forms, at what concentrations across products, or how bioavailable those forms are to the specific bacterial species colonizing the preterm gut. This is a tractable research question with direct translational implications.
Third, consider nickel in the context of nutritional immunity when designing feeding protocols for preterm infants. If the goal is to reduce the activation of nickel-dependent virulence pathways, then minimizing dietary nickel during the window of maximum vulnerability (before the infant's own nutritional immunity system is functional) is a logical intervention. This does not require new drugs. It requires attention to a metal that no one is paying attention to yet.
The broader question
Every parent who buys infant formula trusts that it has been tested for things that could harm their baby. Heavy metal testing for infant food is expanding, driven by regulatory pressure from the FDA's Closer to Zero initiative and consumer advocacy. But that testing currently focuses on lead, arsenic, cadmium, and mercury.
Nickel is not on the list.
Given what we know about nickel-dependent virulence factors, about the preterm gut's vulnerability, and about the mechanism by which dietary nickel could activate the very bacteria that cause NEC, the question is not whether nickel should be tested. The question is why it isn't tested already.
I don't have a satisfying answer to that question. But I do have a certification framework that includes nickel, and I intend to make sure that every infant food brand that wants to demonstrate they take heavy metals seriously has the option to test for all eight metals, not just the four that regulators currently prioritize.
If you're a food brand, a formula manufacturer, or a researcher working on NEC, I want to hear from you. Get in touch.